What is Titration?
A titration is a chemical analysis in which a researcher determines the concentration of a chemical solution (analyte or titrand) by adding a measured volume of standard solution (titrant) until the produced reaction reaches the equivalence point. Titrations are generally used to find the unknown concentration of an acid or base in solution.
Traditional Titration Procedure
To perform a titration, clamp an empty burette above a beaker or an Erlenmeyer flask. Carefully fill the burette with a titrant of known concentration, and fill the flask with a known volume of the analyte of unknown concentration. Add a magnetic stir bar to the analyte solution to assist with mixing. Then, adjust the stopcock on the burette, and slowly drip the titrant into the analyte.
 Add a pH Sensor, chemical indicator, or both to the analyte solution to assist with tracking the titration’s progress. During the titration,
the pH changes as the titrant is added. Plotting the analyte pH against the titrant volume produces a graph known as a titration curve.
Add a pH Sensor, chemical indicator, or both to the analyte solution to assist with tracking the titration’s progress. During the titration,
the pH changes as the titrant is added. Plotting the analyte pH against the titrant volume produces a graph known as a titration curve.
The shape of the titration curve is analyzed to determine the equivalence point of the reaction. The total titration volume is determined by reading the change in titrant volume from the burette at the equivalence point.
Teaching Tools
Wireless pH Sensor
Monitor pH during chemical reactions, titrations, water quality studies, and more with the Wireless pH Sensor! Transmit your data wirelessly to your device to view pH changes in real-time, or store data for days for long-term studies. A simple coin cell battery that lasts 2-3 years powers the sensor, so it’s always ready for use in your lab!
Modern Titration Procedure
If you’ve ever used the methods described above, then you likely know how tricky acid-base titrations can be. One moment your analyte looks clear, and the next it’s bright fuchsia. For this reason, many modern acid-base titration setups utilize both a pH Sensor and a drop counter so that the titration curve can be plotted and monitored in real time. With this method, the experimenter uses a live plot to predict the equivalence point before it occurs and adjusts the drop rate as it nears for greater precision.
Teaching Tools
Generate Live Titration Curves with the Wireless Drop Counter
The Wireless Drop Counter improves the efficiency of acid-base titrations by automatically recording the volume of titrant added.
Simply align the drop window with the burette, connect the sensor to your device, and begin monitoring the titration in real time. A built-in IR filter protects the drop window from light interference for reliable accuracy, regardless of drop size or pacing.
When used with a Wireless pH Sensor, the Drop Counter generates a live titration curve as the reaction progresses, making it easier to predict both the equivalence point and the endpoint. Titrant volumes can be reported in drops or mL.
Chemical Indicators
A chemical indicator is a substance that visually indicates the presence or absence of a chemical compound, usually by changing color. Most chemical indicators are weak organic acids or bases that exhibit different colors in their protonated and deprotonated states.

Prior to starting a titration, add a few drops of phenolphthalein into the analyte solution. If the solution is acidic or neutral, the phenolphthalein will appear colorless.
Common titration indicators include:
- Phenolphthalein
- Bromothymol Blue
- Methyl Red
- Universal Indicator
- Methyl Orange
Phenolphthalein
Phenolphthalein is a widely used indicator. It is naturally colorless but turns pink when present in a basic solution with a pH of about 8.3 to 10. Above a pH of 9, the fuschia color darkens remarkably; above a pH of 10, the solution becomes colorless once more.
Phenolphthalein is commonly used in acid-base reactions where the analyte neutralizes the mixture, leading to neutrality—the moment just after all the acid and base have reacted. After neutralization is reached, even one or two additional drops of titrant will raise the pH to between 8 and 10, causing the analyte solution to turn pink. Since the indicator changes color almost immediately after neutralization, it serves as a great benchmark for identifying the equivalence point.
Other visual indicators include bromothymol blue, which turns blue between a pH of 6 and 8, or methyl red, which turns reddish-orange between pH 4 and pH 6.
What Causes an Indicator to Change Color During a Titration?
Indicator color change is caused by changes in chemical structure that occur as the indicator binds or releases a proton in solution. You can learn more about the relationship between chemical structure, light, and color in our guide, "What is Spectroscopy?"
Indicators are generally used in low concentrations so they do not significantly alter a reaction’s equivalence point. Typically, the indicator should be a lower concentration than the acid or base being titrated, so it doesn’t interfere with the chemical reaction.
The indicator used depends on the type of titration being performed. For example, there are four types of titrations that involve acids and bases—and they all have different equivalence points. We will discuss equivalence points and titration curves in more detail in the next section.
How to Find the Equivalence Point
The equivalence point of a reaction occurs when the number of moles of the titrant is chemically equivalent to the number of moles of the analyte.
The equivalence point happens immediately before the chemical indicator changes color, as the color change signifies the endpoint of the reaction. A single titration, such as a polyprotic acid-base titration, can have multiple equivalence points but only one endpoint, depending on the indicator involved.
Steps to find the equivalence point:
- Monitor the pH of the analyte with a pH probe.
- Monitor the reaction visually with an indicator.
- Analyze the titration curve.
We can calculate the equivalence point for an acid-base titration using just a few values. Recall that $ M = \Large \frac {mol}{V} $ and that at equivalence there are equal moles of both hydrogen and hydroxide ions.
$ Mole \: H^+ = Mole \: OH^- \: where \: Mole \: H^+ = H Mole \: H_nA \: for \: diprotic \: (2x) \: or \: triprotic \: (3x) \: acids $
$ \large M_{H+} V_{acid} = \large M_{OH-} V_{base} $
We can use stoichiometry to determine the amount of hydroxide ions (OH-) required to neutralize the hydrogen ions (H+). In the neutralization equations above, the n subscript comes from the H+ and OH– subscripts included in the balanced chemical equation. By measuring the exact volume of the reagent required to reach the equivalence point, it is possible to calculate the molarity of the unknown solution. So the above equations become:
$ \large M_{H+} = \Large \frac {M_{OH-} \: \times \: V_{base}}{V_{acid}} $
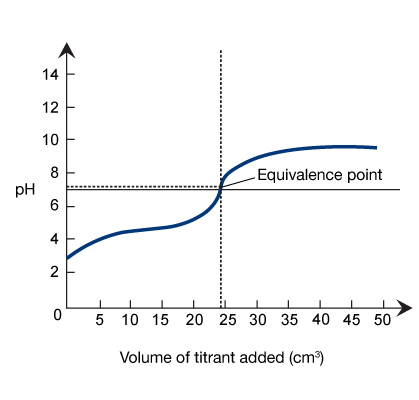
Titration Curves
A titration curve graphically represents the changes in pH and volume that occur during a titration, making it easier to identify the equivalence point of a reaction. It is a plot of the pH of the analyte versus the volume (mL) of titrant added during the reaction.
The shape of a titration curve is used to track the reaction’s progress. As the equivalence point nears, the curve slopes upward, and the pH changes more drastically. After the equivalence point, the endpoint is reached, and the curve begins to flatline. For an acid-base titration, the shape of the titration curve depends on the type of acid and base used, their concentrations, and the volume of titrant added. Keep reading to learn how to interpret common types of titration curves.
Get the Lab

Titration Curves Lab (Free Download)
Discover how titration curves differ between strong and weak acid solutions and determine the equivalent point for each by following our Titration Curves Lab. Your students will get to be chemists when they use a pH sensor and drop counter to make measurements and calculate unknown concentrations!Acid-Base Titrations
The objective of an acid-base titration is to solve for the unknown concentration of an acid or base by performing an acid-base reaction. Acid-base titrations have a wide range of applications in a variety of industries, ranging from pharmaceuticals to food quality assurance. There are four types of acid-base titrations: Strong acid-strong base, weak acid-strong base, strong acid-weak base, and weak acid-weak base. In the next section, we’ll explain the different reactions and how to interpret their corresponding titration curves.
Strong Acid-Strong Base Titrations
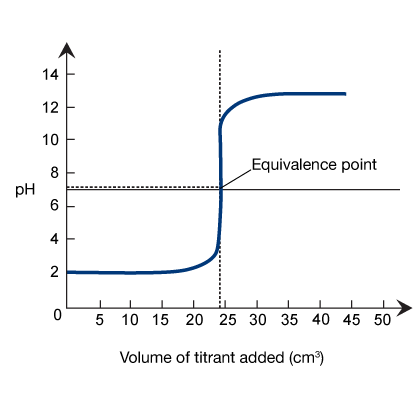
When titrating a strong acid with a strong base, the reactants are highly dissociated, and the pH changes rapidly as the equivalence point nears.
The equivalence point marks the moment at which just enough titrant has been added to neutralize the analyte solution. In a strong acid-strong base titration, the equivalence point of the reaction occurs when the analyte solution reaches a pH of 7.
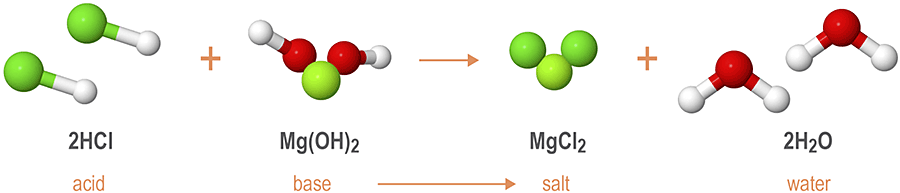
Reactions between strong acids and strong bases are also known as neutralization reactions, as both species fully dissociate to form the neutral products of salt and water, as shown below.
In the curve below, the strong base, NaOH, titrates the strong acid, HCl. As the total amount of added NaOH increases, the pH of the analyte solution also increases.
$ \large NaOH (aq) + HCl (aq) → NaCl (aq) + H_2O (l) $
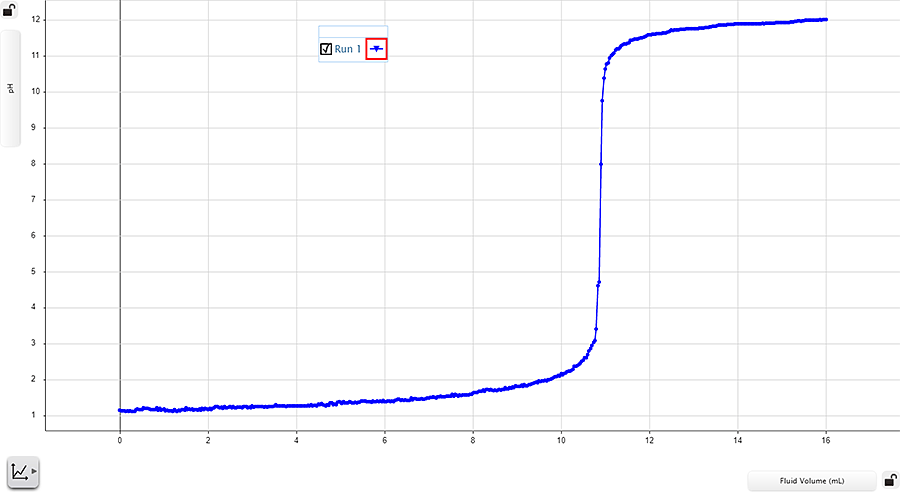
Notice how the curve transitions from strongly acidic (below pH 2) to strongly basic (above pH 11). As the hydroxide (OH–) ions neutralize the hydronium (H+) ions, the curve takes a vertical shape. It then begins to flatten as the number of free hydronium ions decreases and the analyte pH becomes basic.
In any acid-base titration, the equivalence point occurs at the midpoint of the vertical portion of the titration curve.
Strong Base-Strong Acid Titrations
When the solutions have opposite characteristics (basic analyte/acidic titrant), the titration curve is inverted; meaning the curve begins high (basic) and slopes downward toward the low pH range (acidic) as the reaction progresses.
Get the Lab

Investigation of Acid-Base Titrations (Free Download)
Compare the pH titration profiles of strong, weak, and polyprotic acids with our free lab, Investigation of Acid-Base Titrations.Strong Acid-Strong Base Example | Titration of HCl with NaOH
To better understand strong acid-strong base titrations, let’s consider an example. Suppose we want to determine the concentration of hydrochloric acid (HCl) using sodium hydroxide (NaOH) as a titrant. We know that sodium hydroxide is a strong base and hydrochloric acid is a strong acid.
First we would obtain a known volume of hydrochloric acid and titrate it with sodium hydroxide until we reach the equivalence point. This point is determined using a visual indicator, a pH probe, or both. For the titration of a strong acid and strong base, the equivalence point happens at a pH of 7, so we could use Bromothymol blue or Phenolphthalein as the indicator.
After the titration, we can use the balanced chemical equation plus the known volume and concentration of the sodium hydroxide to calculate the unknown concentration of the hydrochloric acid.
Calculating the Unknown Molarity
Imagine the titration involves titrating 25 mL of HCl with 0.10 M NaOH. We want to determine the molarity of the HCl in the 25 mL solution.
We titrate the HCl with the NaOH and find that it takes 55 mL of NaOH to reach the equivalence point.
Now we can calculate the moles of NaOH used in the titration by converting the volume to liters and multiplying it by the known molarity.
$ \large (55 \: mL \: NaOH)(\frac {L}{1000 \: mL})(\frac {0.10 \: mol \: NaOH}{L \: NaOH}) = 0.0055 \: moles \: NaOH $
In the balanced equation below, we can see that the reaction between HCl and NaOH has a 1:1 mole ratio. Therefore, we know that there will be equal moles of HCl and NaOH at the equivalence point. We can now find the molarity (M) of HCl by dividing the number of moles by the known volume of HCl.
$ \large HCl + NaOH \rightarrow H_2O + NaCl $
$ \large (25 \space mL \space HCl)(\frac {L}{1000 \space mL}) = 0.025 \space L \space HCl $
$ \large (\frac {0.0055 \space mol \space HCl}{0.025 \space L \space HCl}) = 0.22 \space M \space HCl $
When the mole ratio is 1:1 for an acid-base titration no differentiation is needed to compare acid concentration and H+ concentration. We can calculate the unknown concentration using the equation below, where M is molarity (M) and V is volume:
$ \large M_AV_A = M_BV_B $
$ \large (M_A)(0.025 \: L \: HCl) = (0.10 \: M \: NaOH)(0.055 \: L \: NaOH) $
$ \large M_A = 0.22 \: M \: HCl $
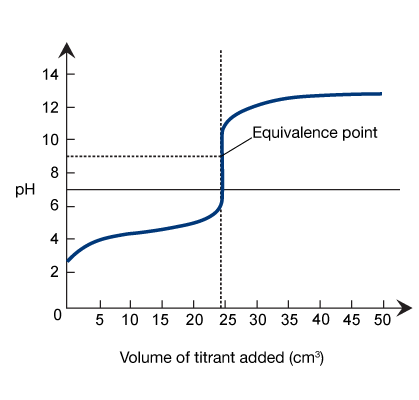
Weak Acid-Strong Base Titrations
In a titration between a weak acid and a strong base, only the strong base dissociates fully. Once the equivalence point is reached, the excess hydroxide ions accumulate in the analyte solution, and the titration curve shifts toward strongly basic. For these types of reactions, the analyte solution is considered basic at the equivalence point (pH > 7).
 The initial pH of the analyte in a weak acid-strong base titration is higher than the initial pH of the analyte in a strong acid-strong base titration, because
weak acids only partially dissociate in solution.
The initial pH of the analyte in a weak acid-strong base titration is higher than the initial pH of the analyte in a strong acid-strong base titration, because
weak acids only partially dissociate in solution.
Get the Lab

Weak Acid Titration Lab (Free Download)
In this lab, students will use a drop counter and pH sensor to monitor monoprotic and polyprotic weak acid titrations. They’ll use the data they collect to determine the relationship between the shapes of their titration curves and Ka.Strong Acid-Weak Base Titrations
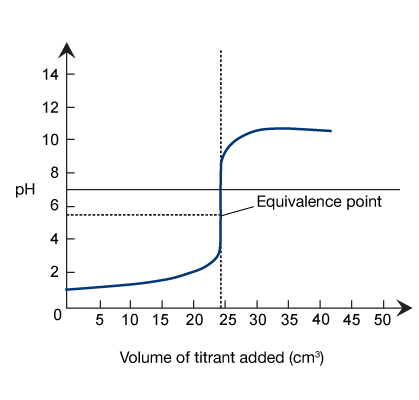
In a titration between a strong acid and a weak base, only the strong acid dissociates fully. At the equivalence point, the analyte solution contains a slight excess of hydronium ions, which keep the pH of the analyte solution below 7.
 In a strong acid-weak base titration, the starting pH of the analyte is lower than the initial pH of the analyte in a strong
acid-strong base titration. This is because weak bases dissociate less than strong acids.
In a strong acid-weak base titration, the starting pH of the analyte is lower than the initial pH of the analyte in a strong
acid-strong base titration. This is because weak bases dissociate less than strong acids.
Weak Acid-Weak Base Titration | Titration of Acetic Acid with Ammonium Hydroxide
In a titration between a weak acid and a weak base, both the analyte and the titrant dissociate partially. At the equivalence point, the analyte approaches neutralization (pH = 7). For these reactions, the initial pH of the analyte generally ranges from three to eleven. The titration curve resembles a compressed version of the traditional “S” shape, and the equivalence point is indicated by the smaller but still steep rise in the curve.
For example, consider the following equation between acetic acid (a weak acid) and ammonium hydroxide (a weak base):
$ \large CH_3COOH(aq) + NH_4OH(aq) ⇌ CH_3COONH_4(aq) + H_2O(l) $
 In a weak acid-weak base titration, neither the analyte nor the titrant dissolve fully in solution. The resulting curve is a
compressed version of the strong acid-strong base titration curve.
In a weak acid-weak base titration, neither the analyte nor the titrant dissolve fully in solution. The resulting curve is a
compressed version of the strong acid-strong base titration curve.
Comparing Titration Curves
The plot below shows the titration curves produced from two different titrations: a weak acid-strong base titration and a strong acid-strong base titration.
Reaction 1 (Strong Base-Weak Acid): The blue line is the titration curve produced when the strong base, sodium hydroxide (NaOH), titrates the weak acid, acetic acid (CH3COOH).
$ \large CH_3COOH(aq) + NaOH(aq) ⇌ Na^+CH_3COO^–(aq) + H_3O^+(aq) $
Reaction 2 (Strong Base-Strong Acid): The red line is the titration curve produced when the strong base, ammonium chloride (NH4OH), titrates the strong acid, hydrochloric acid (HCl).
$ \large HCl(aq) + NH_4OH(aq) → NH_4Cl(aq) + H_2O(l) $
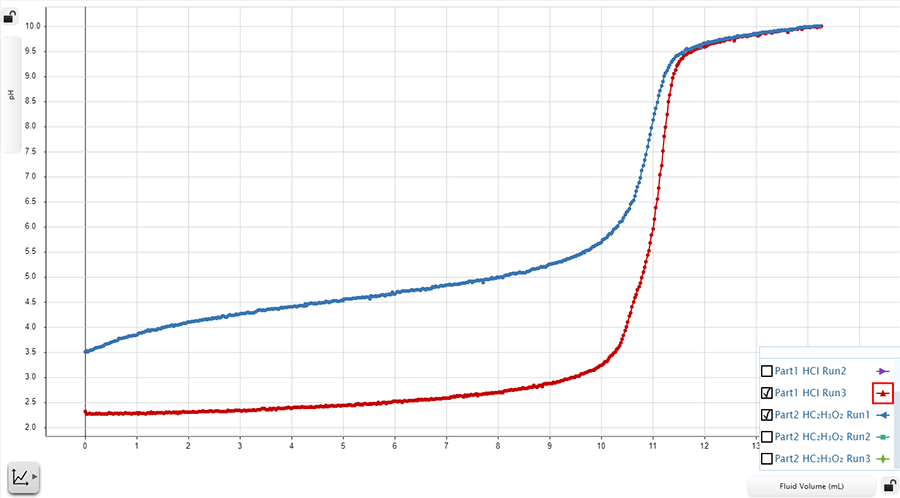 A strong base-weak acid titration (blue) is plotted against a strong base-strong acid titration (red).
A strong base-weak acid titration (blue) is plotted against a strong base-strong acid titration (red).
In the plot above, the analyte solution in Reaction 1 begins at a higher pH than the analyte used in Reaction 2. Weak acids generally have pH values of about 7. Reaction 1 uses the weak acid, acetic acid, which only partially dissociates. In contrast, Reaction 2 utilizes the highly dissociative, strong acid, hydrochloric acid, as the analyte. Reaction 2 uses a more dissociative acid, and thus, has a higher concentration of hydronium ions in the analyte solution, giving it a lower pH.
Because the analyte in Reaction 1 doesn’t fully dissociate, the addition of the highly dissociative strong base, sodium hydroxide, quickly neutralizes the acid in solution. At the equivalence point for Reaction 1, the pH of the analyte is greater than seven. This is true for all titrations that involve a weak acid and a strong base.
In Reaction 2, both the analyte solution and the titrant fully dissociate because it is a strong acid-strong base reaction. At the equivalence point, the analyte solution contains chemically equal amounts of hydroxide and hydronium ions, and the pH is near 7.
Teaching Tools
Log, Plot, and Monitor Temperature Data on Virtually Any Device!
With its waterproof design, long-lasting battery, and live results, the Wireless Temperature Sensor is the perfect tool for tracking temperature in the classroom, lab, or field. Pair it to your device via Bluetooth to monitor measurements in real time, or log data to the onboard memory for analysis later. Explore freezing and melting points, measure endothermic and exothermic reactions, monitor titrations, and much more!
Polyprotic Acid-Base Titration Reactions
While most acid-base reactions have a single equivalence point, polyprotic acids and bases produce reactions with multiple equivalence points. A polyprotic acid can donate more than one proton in solution, while a polyprotic base can accept more than one proton in solution.
When used in a titration, the protons in a polyprotic acid ionize in succession, resulting in multiple equivalence points. A similar effect is seen when using polyprotic bases, which accept protons in succession. On a titration curve, the multiple equivalence points occur at the middle of each vertical section.
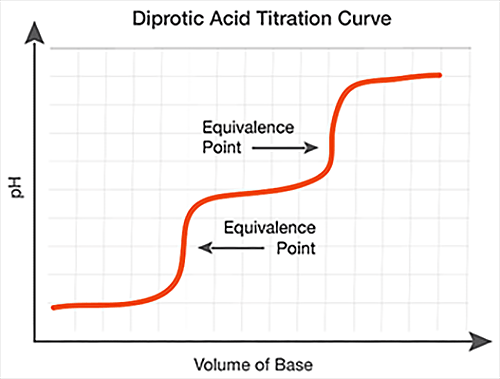
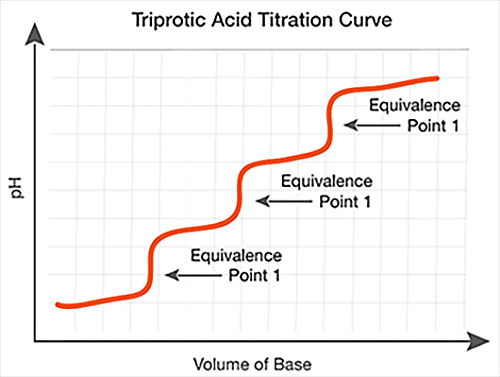 Polyprotic acid titrations have multiple equivalence points, as shown in the graphs above. The equivalence points are located in the middle of each vertical section.
Polyprotic acid titrations have multiple equivalence points, as shown in the graphs above. The equivalence points are located in the middle of each vertical section.
Examples of polyprotic acids include the diprotic acid, sulfurous acid (H2SO3), which donates up to two protons per molecule and the triprotic acid, phosphoric acid (H3PO4), which has three dissociable protons.
Examples of polyprotic bases include the diprotic base, sulfate ion (SO42-), which accepts up to two ionized protons, and the triprotic base, phosphate ion (PO43-), which accepts up to three ionized protons.
Other Types of Titrations
Acid-base titrations are just one type of titration used by chemists to determine the unknown characteristics of a solution. Other types of titrations include redox titrations, conductometric titrations, precipitation titrations, complexometric titrations, and non-aqueous titrations.

Discover more than a dozen standards-based titration labs—all available for free from the PASCO Experiment Library.
Redox Titrations
A redox titration utilizes reduction and oxidation reactions to determine the concentration of a known analyte. Whereas acid-base titrations follow the exchange of protons, redox reactions follow the exchange of electrons. In a reduction reaction, electrons are gained. In an oxidation reaction, electrons are lost; the other added substance would gain these lost electrons and become reduced. Reduction and oxidation reactions always occur in pairs, hence the term “redox reaction.”
In the following redox titration, iron (II) is oxidized, and chromium (VI) is reduced. Fe2+ loses an electron and becomes Fe3+.
Cr2O72- accepts electrons from the hydrogen ions, producing water and Cr3+.
$ \large 6Fe^{2+} + {Cr_2O_7}^{2–} + 14H^+ → 6Fe^{3+} + 2Cr^{3+} + 7H_2O $
Redox titrations are often measured using a special probe, called an oxidation reduction potential probe (ORP Probe). ORP probes measure the oxidative and reductive potential of an analyte solution, enabling redox reactions to be tracked in real time.
Teaching Tools
Oxidation Reduction Potential Probe
The Oxidation Reduction Potential Probe connects to the Wireless pH Sensor and allows students to determine the ability of a species in a solution to act as an oxidizing agent or reducing agent during redox reactions. Use this probe to monitor solutions during oxidation-reduction titrations, perform water quality studies, and study the effects of water chlorination. This probe is not a standalone sensor. It connects to and requires an amplifier.
Conductometric Titrations
In a conductometric titration, the concentration of the analyte solution is determined by tracking the exchange of ions during the reaction. To do so, the conductivity of the analyte is measured throughout the procedure, usually with a conductivity probe such as the Wireless Conductivity Sensor.
Get the Lab

Stoichiometry in Solutions Lab (Free Download)
Investigate conductometric titrations with your students with our Stoichiometry in Solutions Lab! Download the full lab for free.Why is Conductivity Used in Titrations?
Conductivity is a measurement of an ion’s capacity to conduct electricity in solution, also known as its ionic or electrolytic conductivity. During a titration, the reagents dissociate into ions, each of which has a specific ionic conductivity. When one ion replaces another, the conductivity of the analyte solution changes, making it possible to estimate the reaction’s progress.
The conductivity of a solution depends on the concentrations of cations and anions present and the temperature of the solution. To a smaller extent, conductivity also depends on an ion’s charge and mobility. When larger, less mobile ions, such as a sodium ion, replace a mobile ion, such as a hydrogen ion, the conductivity of the solution decreases. This effect is larger in more viscous fluids.
Teaching Tools
Wireless Conductivity Sensor
The Wireless Conductivity Sensor measures the electrical conductivity of an aqueous solution. It is ideal for investigating the properties of solutions, including total dissolved solids (TDS) for water quality inquiry. Because it is temperature compensated, calibrations are less frequent and can be applied across a range of temperatures. With a range of 0 to 20,000 µS/cm, this sensor can be utilized for chemical, biological, and environmental studies.
Covalent and Ionic Compounds
Covalent and ionic compounds make excellent reactants for conductometric titrations because they exhibit different ionic conductivities. A covalent compound cannot conduct electricity in its solid, liquid, or molten state. In contrast, an ionic compound can conduct electricity, so long as it is dissolved in solution or used in its molten form, and its ions are free to move.
- Water (H2O)
- Carbon monoxide (CO)
- Carbon dioxide (CO2)
- Ammonia (NH3)
- Methane (CH4)
- Sodium chloride (NaCl)
- Sodium carbonate (Na2CO3)
- Calcium carbonate (CaCO3)
- Lithium chloride (LiCl)
- Calcium Iodide (CaI2)
Precipitation Conductometric Titration
In a precipitation conductometric titration, an electrolyte titrates another electrolyte, forming an insoluble product called a precipitate. Precipitation titrations follow the general solubility rules.
During a precipitation titration, the conductivity of the analyte solution usually drops with the loss of ions as they form precipitate. The conductivity continues to drop until the endpoint is reached. After the endpoint, adding additional titrant adds more ions to solution. The conductivity rises until it matches the conductivity of the titrant, and then it remains constant.
One example is the precipitation reaction between sodium carbonate Na2CO3 and calcium chloride CaCl2.
$ \large Na_2CO_3 + CaCl_2 → NaCl + CaCO_3 $
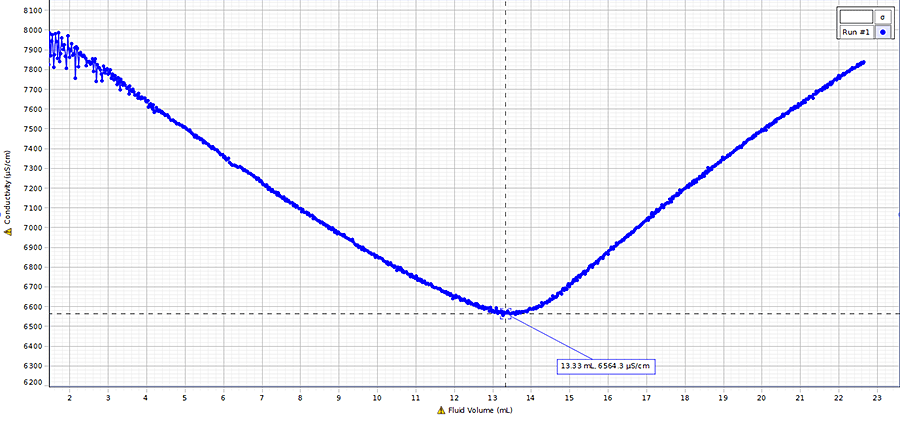 Conductivity of the analyte solution is plotted against the volume of added titrant for precipitation conductometric titrations.
Conductivity of the analyte solution is plotted against the volume of added titrant for precipitation conductometric titrations.
When sodium carbonate titrates calcium chloride, the calcium (Ca2+) ions and the carbonate (CO32-) ions precipitate out and (Na+) replaces them. On the titration curve, the portion of the curve sloping downward indicates this exchange. The reaction equivalence point occurs once all the ions have been exchanged, indicated by the lowest point on the curve. The reaction begins to reverse when (Na2CO3) is added beyond the endpoint, increasing the conductivity. The curve will eventually level out and become a flat line once the ion levels of the solution are equal to the ion levels of the titrant.
When a strong base, such a NaOH, is used in a conductometric titration, the conductivity of an analyte with two acids—one weak acid and one strong acid—can be determined.
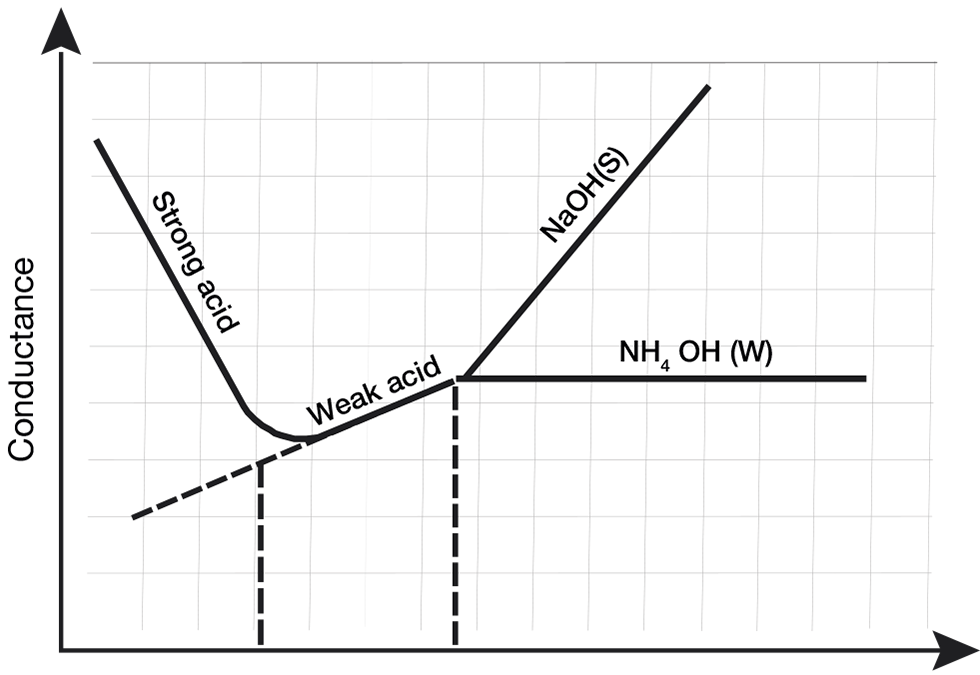 This graph shows the conductivity change when the strong base NaOH is added to a solution of both strong and weak acids.
This graph shows the conductivity change when the strong base NaOH is added to a solution of both strong and weak acids.
The strong acid is neutralized first, as indicated by the steep downward curve as ions form water and salt. However, the weak acid neutralization encourages more acid to dissociate until the acid is neutralized. After this equivalence, conductivity will rise quickly due to the added strong base. The conductivity due to the conjugate weak acid will no longer change as no more is being produced and is shown as a straight line in the graph.
Non-Aqueous Titrations
In a non-aqueous titration, a solvent other than water dissolves the analyte substance. The amphoteric properties of water make it a tough competitor for very weak acids and bases, which is why non-aqueous titrations exclude it from the reaction.
Non-aqueous acid-base titrations are used when:
- the species of interest is not soluble in water
- the sample contains fats or oils
- the sample contains a mixture of acids or bases that must be identified by titration
- both the titrant and the analyte have very weak basic or very weak acidic properties
Commonly used non-aqueous solvents include:
- ethanol
- methanol
- isopropanol
- toluene
- glacial acetic acid
Other Types of Titrations
Titration is a widely used analytical technique in chemistry to determine the concentration of a substance in a sample. There are many different types of titration methods, each with its own unique advantages and limitations. Learn more about the different methods of titration in our blog, Beyond Acid-Base: Other Titration Types.